Covid-19 & Vitamin D : demonstration of proof
The evidence that a low blood level of vitamin D leads to an increased risk of serious or fatal Covid-19 infection, compared to those with higher (ideal) levels, is indisputable. There is widespread deficiency of vitamin D and this is a world-wide phenomenon. 47% of the population pf Spain are deficient in vitamin D, and 80% of those admitted to hospital with Covid-19.
There are several reasons for this widespread deficiency, including extensive skin cover by clothing, many more people working indoors, and in the age of air conditioning many people seeking leisure indoors where it is cooler. The problem is compounded by much of the population living in the temperate zones of the northern hemisphere where the sun produces vitamin D in the skin for only six months of the year. This puts people with melanin-rich skin living in temperate zones at a great disadvantage, as melanin blocks much of the UV light and diminishes vitamin D production. The atmospheric pollution of world cities blocks UV light penetration to ground level and also reduces vitamin D production.
Low blood levels of vitamin D associated with severe or fatal Covid-19 have been demonstrated in India, Singapore, Newcastle upon Tyne, Iran, Turkey, Mexico, France and St Petersburg. Low blood levels of vitamin D predicting Covid-19 have been demonstrated in Bari (severe or fatal), Chicago, Israel, Santander, Heidelberg (severe or fatal), Boston, and Tehran (severe or fatal).
The benefit of treating with Covid-19 pneumonia with vitamin D patientshave been demonstrated in Singapore and Córdoba, Spain. The latter was a randomised controlled trial, using Calcifediol, the part-activated form of vitamin D.
But the government and its advisors deny that vitamin D should be used as a prevention or treatment of Covid-19. A fact that is hard to believe is that the advisory body NICE (National Institute for Health and Care Excellence) has advised that the very positive study from Córdoba should not influence the way in which patients with Covid-19 pneumonia are to be treated. This is outrageous.
It is claimed that there is an absence of "proof"
When most people use the word "proof" they only rarely understand the meaning of the word. When challenged, they will usually say "Well, you know, er...." But is essential to understand what it means.
Proof is "The fulfilment of pre-determined criteria". In other words the question of proof must be thrown back at the sceptic. He or she must decide the criteria that would be accepted as proof of a given proposition. Proof would be based on the evidence that would lead to the recognition of truth. The criteria must be testable, real science.
The problem is that there is no absolute truth, and the evidence of proof is always pragmatic. New evidence might bring about a re-evaluation of truth. For example the proposition that the sum of the angles of a triangle is always 180 degrees was accepted until the 20th century when the triangle was superimposed on the curvature of the Earth.
The important decision in reaching the truth is based on the legal concept of the weight of evidence, but then there is a problem. Karl Popper used the example of swans, in which a proposition might be made that all swans are white. Researchers might travel widely counting white swans thereby adding to the "weight of evidence". But then a black swan is discovered. The truth of the proposition does not become determined by the majority. The weight of evidence might be in favour of swans being being white, but the single black swan invalidates the assertion. Popper championed falsification (the null hypothesis) as the true pursuit of experimentation,
We must be careful. The propostion that low levels of vitamin D lead to a increased incidence of critical or fatal Covid-19 is not quite so black and white as swans.
It might be found that some people with what are considered to be ideal blood levels of vitamin D might become critically ill and die from Covid-19. We know that some people have a genetic inheritance of sub-optimal vitamin D receptors (VDR), called polymorphisms. This is has been recognised in the clinical syndrome of "vitamin D resistant rickets", in which vitamin D does not work. Its target molecule (VDR) is defective and so the key does to fit in the lock. This has been found to be relatively common in people with tuberculosis, leading to sub-optimal immunity.
But it is rare and we must look at the great majority when seeking proof, accepting exceptions.
QED
An experiment must always have controls, and I first learnt this in science at school. If we want to investigate the proposition that the growth of a seed requires the presence of water, we put two seeds in two pots of compost and apply water to just one. The criterion of proof is that all other things being equal only the seed exposed to water will grow.
As in mathematical solutions, we would then state "QED", quod erat demonstrandum, that has been demonstrated, proved.
Simple experiments in humankind are usually not quite so straightforward, but they can be. Correction of deficiency states are easy to understand once that the deficiency is understood by measurement of a physiological parameter. It became clear that Type 1 Diabetes is due to deficiency of insulin, the failure of insulin being manufactured in the pancreas. The administration of insulin to someone in a diabetic coma was like a miracle, there having been many unfortunate "historical control" patients who had died before the discovery of insulin (I appreciate that the measurement of insulin in the blood was not possible at the time, but it could be inferred). The clinical experiment of a randomised controlled trial (RCT) was unknown at the time (1922) but it might be enforced today. Similarly deficiency of thyroid hormone and its replacement in treating myxoedema, or cortisone in Addison's disease.
Asleep at the wheel
During the pandemic of Covid-19 there has been a refusal rather than a reluctance to consider vitamin D deficiency as a major risk factor for critical and potentially fatal disease. There has been in the UK no co-ordinated effort to investigate the extent of vitamin D deficiency, no effort to associate it with the level of severity of Covid-19, and no attempt to provide vitamin D replacement therapy for those deficient. The evidence of 18 published studies has been ignored, and there has been no scientific undertaking to disprove the proposition.
The UK has witnessed almost 50,000 deaths, and yet the government or its advisors have failed to provide a very cheap, very safe, and readily available way to minimise deaths.
Sir Austin Bradford Hill and Sir Richard Doll – the importance of careful observation
If lack of proof, or rather a serious lack of understanding of proof, has been paralysing the government and its advisors, let me bring to attention the very important work of the distinguished British statistician Sir Austin Bradford Hill, who I have mentioned in previous Blog posts. Repetition can be essential.
Sir Austin Bradford Hill and Sir Richard Doll produced in 1950 the important study that brought about the general acceptance that cigarette smoking causes lung cancer, using a sample of doctors who declared their smoking habits. It was a time-related (temporal) observational study and the results can be seen in the figure.
It is obvious that the more cigarettes smoked per day, the greater the chance of dying from lung cancer.
Note that this was not a double blind randomised controlled trial. To create an RCT would have been an impossible task. Think about it. It was an observational study, a method discounted by our supposedly betters concerning Covid-19 and vitamin D.
The authors of the study had thought about the meaning of proof, and in 1965 Bradford Hill produced a set of criteria. He considered that if these were met, it would constitute "proof".
We must apply these to the proposition that:"Vitamin D deficiency leads to an increased risk of critical or fatal Covid-19"
The studies quoted are reviewed in a previous Blog post
1. Strength of association:
Examples:
A study from India demonstrated the following association in Covid-19:
Blood vitamin D level >30ng/ml, 62% had severe disease
Blood vitamin D level <30ng/ml, 85% had severe disease
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7276229/
I have mentioned a study from Spain (published on October 27th 2020):
Covid-19 hospital patients: 80% deficient in vitamin D.
Community controls: 47% deficient in Vitamin D.
2. Consistency of association:
Similar associations were demonstrated in:
3. Specificity:
The studies that I have listed are specific for the link between low levels of vitamin D and high risk of critical illness or death from Covid-19. However I accept that not everyone with a low blood level of vitamin D will become seriously ill or die from Covid-19 infection. Similarly, not everyone who dies from Covid-19 is deficient in vitamin D. There are other factors at work in a sub-optimal immune system (for example VDR polymorphisms), but it is not possible to measure "immunity" directly.
4. Temporality:
The association between cigarette smoking and lung cancer was criticised on the basis that having lung cancer might cause people to having smoked cigarettes decades earlier. This is called "reverse causality" but one the example illustrated it is not very plausible.
It has been stated with some truth that a serious infection with Covid-19 reduces the blood level of vitamin D. I have indicated previously that this is true in that vitamin D (as the active 1,25(OH)D) is consumed, irreversibly inactivated after use. It is therefore essential for the blood level of 25(OH)D not to be low in advance of the infection. A reservoir of 25(OH)D in the blood is essential if a serious infection occurs so that production of 1,25(OH)D can be escalated.
It is important that observations are time-related, the sequence of events being demonstrated rather than just a "snapshot". Temporality means the ability of a low level of vitamin D to predict critical or fatal Covid-19. This has been demonstrated in the following studies:
Bari.
Blood level of vitamin D >30ng/ml, 5% died subsequently
Blood level of vitamin D <10ng/ml, 50% died subsequently
5. Biological gradient:
This criterion is met by looking at a range of blood levels of vitamin D and relating them to the development of or the outcome from Covid-19. This information is best illustrated in the study from Israel, but there are limitations as it looked at ethnicity and infection, and did not include deaths. In respect of Covid-19 infection rates, there appeared to be a threshold level at 30ng/ml, above which infection rates drop to half compared to lower blood levels.
A biological gradient was demonstrated in the Heidelberg study, with unfavourable outcome progressively increasing with decreasing blood vitamin D levels. The hazard ratio, the risk of adverse events, for the lowest vitamin D levels was 14.73 compared to the highest, with a gradient between.
Hazard ratios for death:
Vitamin D >30ng HR = 1.00
Vitamin D <20ng HR = 11.27
Vitamin D <12ng HR = 14.73
The gradient can be seen on the graph. The higher the blood level of vitamin D, the lower the death hazard (>20ng/ml is standardised as 1).
6. Plausibility
Is it biologically plausible that vitamin D might protect against Covid-19 symptomatic infection, the cytokine storm, escalation of illness to a critical, and death? The answer is that it is very plausible, based on a large amount of research that has been undertaken since about 1980. The role of vitamin D is often dismissed on the basis of ignorance of this information.
There is a wealth of published information that demonstrates that vitamin D has a pivotal role in activating VDR (vitamin D receptor), which in turn activates genes that control T- and B-lymphocytes and suppress TNF-alpha. I have described these processes in a previous post in April of this year.
7. Coherence
This involves linking together the information obtained from basic science with information obtained from observations of the population and the Covid-19 pandemic. There is a very coherent story, which includes the serious disadvantage of BAME people (Black and Asian Minority Ethnic) in the UK, US, and Sweden, based on their known low blood levels of vitamin D. This could have been investigated in much more detail during the pandemic, but if that happened ( and I doubt it) the results are not yet released.
The proposition that low blood levels of vitamin D predispose to critical disease and death from Covid-19 is also supported by the observations of a latitude effect, that there have been few deaths in equatorial Africa, and deaths per million are more than 100 times higher in the UK.
8. Experiment
There are strict limitations on experiments that can be performed on humankind, especially when these involve the correction of a demonstrated deficiency state. We find both logistic challenges and also ethical constraints. At a a time of a dangerous pandemic, should people be denied the potential benefits of a known and a safe pre-hormone such as vitamin D, of which they are definitely or likely to be deficient?
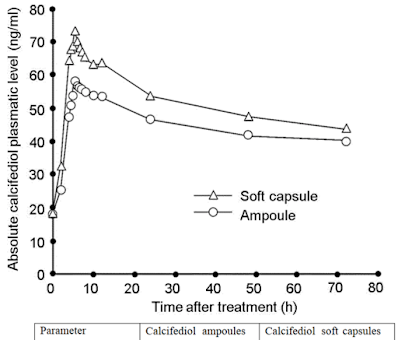
However a clinical experiment (RCT, randomised controlled trial) was undertaken at the Reina Sofía University Hospital, Córdoba, Spain. Of 50 patients with Covid-19 pneumonia receiving standard high quality care plus vitamin D in its part-activated form Calcifediol, just one required transfer to the ICU and there were no deaths. In the randomly selected 26 in the control group, who received standard high quality care only, 13 required transfer to the ICU and two died.
This was an experiment with a very positive result, despite it being rubbished by NICE. Unfortunately two deaths occurred. Had this been a trial of a vaccine or a new anti-viral agent, the results would have made headlines in all newspapers. But with vitamin D this was not to be.
More similar experiments might follow. The problem is that natural vitamin D might be used, but it takes one or two weeks to achieve good blood levels of the liver-activated form 25(OH)D. The Córdoba study used by mouth 25(OH)D which is Calcifediol. This bypasses the slow liver hydroxylation process and achieves good blood levels within just two hours. My concern is that future RCTs will use natural vitamin D and will fail to produce a positive result. It would be the rial structure at fault, not vitamin D.
9. Analogy
Are there any studies that are similar to the identification of the deficiency of a natural hormone being associated with a health disadvantage? Of course there are. We have the recognition of Type 1 Diabetes resulting from insulin deficiency and being corrected by insulin replacement therapy. We have the recognition of the clinical syndrome of myxoedema being the result of under-activity of the thyroid gland, and corrected by thyroid hormone. We have Addison's disease being identified as being the result of failure of the adrenal glands and being corrected by cortisone.
There is the rare VDR polymorphism in which vitamin D fails to activate its target, similar to insulin resistance in which physiological amounts of insulin are not effective (Type 2 Diabetes).
The analogies of the proposition that low blood levels of vitamin D lead to immune deficiency are many and are obvious. Similarly, vitamin C deficiency results in dangerously weak soft tissue
10. Reversibility
If low blood levels of vitamin D are associated with a high risk critical illness or death from Covid-19, does correction of this with vitamin D supplement reverse this risk? Here we have incomplete information.
Unfortunately the study from Córdoba did not measure blood levels of vitamin D. The studies from Bari, Israel, Heidelberg, Boston, Tehran, Santander identified low blood levels being associated with a later unfavourable outcome, but they did not intervene and they did not reverse the low blood levels that they had or could have observed. In Heidelberg and Santander the blood was taken from the patients on admission to hospital but not tested for vitamin D until the end of the trial. This kept the investigators and clinical staff "blind" to the vitamin D status of the patients, and it also avoided any ethical conflict in withholding replacement therapy.
An approach could be to identify on admission to hospital the blood vitamin D levels of patients with Covid-19. The results would be available within one or two hours, and then those with low levels could be randomised to receive vitamin D or just standard care. It is important that vitamin D is given to very ill patients in the form of Calcifediol as explained above. It would be superfluous to randomise between vitamin D and Calcifediol as the different pharmaco-dynamics of them are known. Such an approach of sick patients identified as having vitamin D being denied correction of the deficiency (and informed of this) is most unlikely to receive ethical approval
It is possible that other studies addressing reversibility are awaiting publication, but there is the increasing ethical factor that now all patients (or well people) known to have low blood levels of vitamin D will expect to have this corrected. The effect will be obvious if hospital and ICU admission rates and death rates fall rapidly.
Proof
All the criteria of Sir Austin Bradford Hill have been met, with the possible exception of reversibility, which cannot be assessed ethically.
This means that "Proof" can be accepted.
Need more be said? Is there any reason not to protect people who have a low blood level of vitamin D by correcting it? Is there a reason not to treat with Calcifediol patients sick with Covid-19 pneumonia? If there is it must be demonstrated clearly to the public, ideally in the form of a debate.Since September 3rd when the result of the Córdoba became available, 7344 citizens of the UK have died as the result of Covid-19 (November 8th). When will "proof" be accepted? How many more deaths must occur before the population and the sick are protected using vitamin D?
There is, however, one logistic problem. The nation is not prepared to test the blood levels of vitamin D in the population. It could be achieved but it would take time. However vitamin D in a dose of 4,000 units per day given to all the population without knowing blood levels would be perfectly safe, as identified by SACN, the Scientific Advisory Committee on Nutrition.
Is there sufficient vitamin D to meet the needs of all the population? People such as the BAME, the elderly, the obese, and those with diabetes are known to have a high prevalence of low blood levels of vitamin D and so must have priority. Blood levels of vitamin D could be measured later to check on the appropriateness of dose.
Is there sufficient Calcifediol to meet the needs of of all patients admitted to hospital with Covid-19 pneumonia?
Are preparations being made?
Vitamin D deficiency and Covid-19 : its vital importance in a world pandemic
or from Amazon
eBook from iTunes